An Overview of Fats
1. What is the difference between a saturated and an unsaturated fat?
A fat is a triester of glycerol and fatty acids.
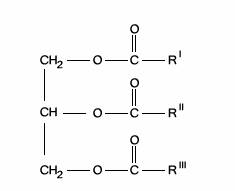
If the fatty acid side chains (R’,R’’and R”’)consist of no double-bonded carbons (C=C), then no more hydrogen can react with it, and the fat is said to be saturated. Such a compound tends to have a higher melting point and is more likely to be solid at room temperature. Fatty acids such as lauric acid, palmitatic acid and stearic acid are all saturated fatty acids. They have 12, 16 and 18 carbon atoms, respectively, with the following formulae:
laurate CH3(CH2)10COO-
palmitate CH3(CH2)14COO-
stearate CH3(CH2)16COO-
(see Biochemistry 3rd Ed by Stryer p 470 for a
more complete list. )
 Saturated
fats have been linked to heart disease. They are a key constituent (40-60%) of
animal fat and of coconut and palm oils. They are also artificially created
through hydrogenation.
Saturated
fats have been linked to heart disease. They are a key constituent (40-60%) of
animal fat and of coconut and palm oils. They are also artificially created
through hydrogenation.
Unsaturated fats have one or more pairs of double-bonded carbons
which can be potentially "saturated" with hydrogen. If only one pair
of double-bonded carbons exists, the fat is said to be monounsaturated. An
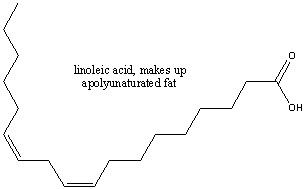
example is oleic acid, a main constituent of olive oil, show below. 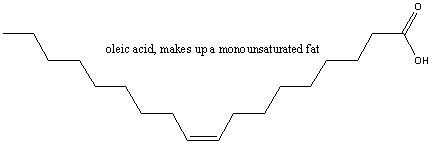 If more than one double-bonded carbons appears then the fat is polyunsaturated. An example
of a polyunsaturated fatty acid is linoleic acid.
Unsaturated fats make up the majority (80-90+%) of
fats found in corn, cottonseed, safflower, soybean and olive oils.
If more than one double-bonded carbons appears then the fat is polyunsaturated. An example
of a polyunsaturated fatty acid is linoleic acid.
Unsaturated fats make up the majority (80-90+%) of
fats found in corn, cottonseed, safflower, soybean and olive oils.
2. Are there any fats which are essential to our diet?
Yes. Mammals cannot introduce double bonds at carbon atoms beyond C-9 in the
fatty acid chain.( Stryer, p
490). Linoleate (18 carbons ,
2 double bonds)
H3C(CH2)4CH=CHCH2CH=CHCH2(CH2)6COO
H3CCH2CH=CHCH2CH=CHCH2CH=CHCH2(CH2)6COO
(Source: Composition of Foods, USDA Handbook No.8 )
Search the
Data Base . Without these polyunsaturated fatty acids, the body
would not, among other things, be able to produce a group of hormones known as prostoglandins, which are needed for regulation of blood
flow to organs, ion transport, modulation of synaptic
transmission and stimulation of inflammation. (Stryer, p992).
Ratio of Linoleic Acid(an omega-6 fatty
acid) to Saturated Fat
|
walnuts |
||||||
|
corn oil |
||||||
|
soybean oil |
||||||
3. Aside from saturated fats, what other fats are unhealthy?
Either to raise the melting point of oils (thus rendering them solid at room temperature) and/or to prolong shelf life, the food industry often hydrogenates unsaturated fats and produces saturated ones. If they partially hydrogenate vegetable oils in order to create a softer product, they create an even greater percent of trans fats through isomerization. These, unlike cis fats, have hydrogen atoms positioned diagonally across a carbon double bond.
More importantly , a study published in Nov. 1997 in the New England Journal of Medicine tracked 80 000 nurses for 14 years, (also see Market Place, January 6th , 1998; and Trans Fat Fact Sheet) and by comparing dietary data, the researchers concluded that trans fats were more likely to lead to heart disease than saturated fats. Whereas saturated fats increase both HDL (high-density lipoproteins, which carry cholesterol back to cell membranes) and LDL (low-density lipoprotein which carry cholesterol to blood vessels), trans fats raise LDL and lower the beneficial HDL. LDL contributes to heart disease by laying down fatty deposits in the walls of arteries.
Even healthy oils can be converted to trans fats through deep frying, specifically when temperatures exceed 180 oC.
For more updated details on trans fats click either here or here.
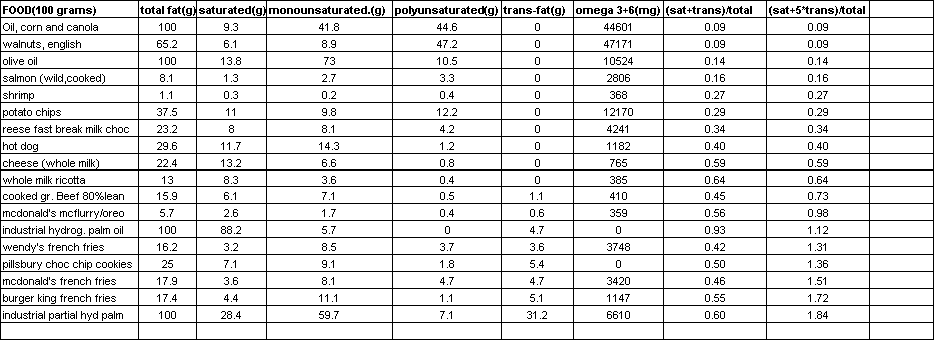
In the table below I selected foods from various food groups and I sorted them according to their ratio of the sum of saturated and trans-fats to their total fat content. In the last column of the spreadsheet, I actually multiplied the number of trans-fat grams by 5 since research suggests that they are worse than saturated fat. The lower the ratio, the healthier the food. Not surprisingly we see corn oil, walnuts and olive leading the way, and the worst fat sources are McDonald’s and Burger King French fries and foods rich in partially hydrogenated palm oil. (Note that some fats are counted twice since omega 3 and 6 are a type of polyunsaturated fat)
“In this large, prospective study of women, we found that a higher dietary intake of saturated fat and trans unsaturated fat was associated with an increased risk of coronary disease, whereas a higher intake of monounsaturated and polyunsaturated fats was associated with a decreased risk.”
-![]() November 20, 1997
November 20, 1997
3. What
is ALA
We have already mentioned linolenate, the
essential fatty acid. If you look at its structure, the acidic H+1 is
missing from the carboxylate group (COO-1).
With the acidic H+1, linolenate becomes linolenic acid, which comes in two forms, the alpha(ALA)
and gamma form. The latter has double bonds in diffferent
positions. The location of the three double bonds of alpha linolenic
acid was shown in the structure of linolenate.
Anyhow, what's important is that ALA has been associated with a lower incidence
of heart disease and stroke. (see Leaf A and Weber PC. N Engl
J Med. 1988;318:549-557 and Dupont J, et al. Am
J Clin Nutr.
1996;63:991S-993S.) A high concentration of ALA in the blood probably prevents
blood clots from forming. Foods rich in ALA are not common but include walnuts,
purslane (flaxseed), canola oil and soybean oil.
Note that hens whose diets are supplemented with flaxseed lay eggs with higher
concentrations of ALA.